
Discover biological insights and / or develop drugs for targets
Discovery‑based biotech platforms use a repeatable engine (novel assays + automation/ML + proprietary data) to discover novel targets and/or develop drug candidates. They monetize via partnerships and an internal pipeline.
Millennium, Vertex, Isomorphic, DESRES, Relay, Schrödinger, Recursion, NewLimit
Compound portfolio companies Achira, Juvena
Discovery Platforms
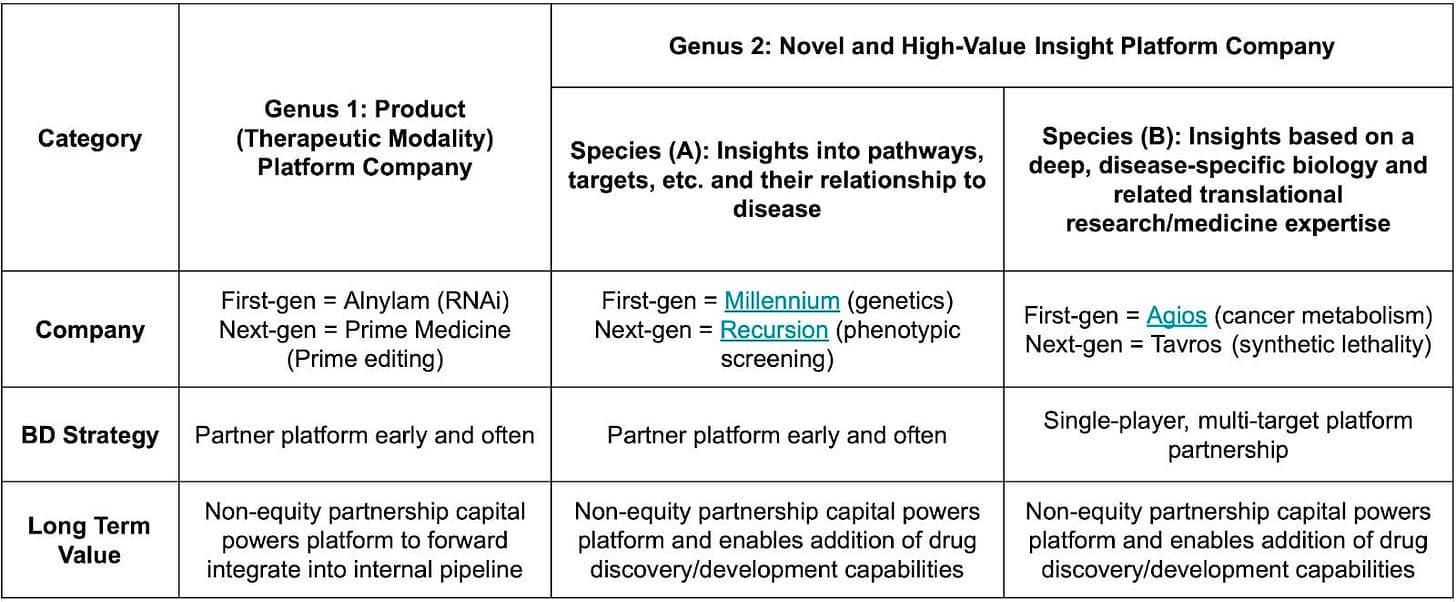
Discovery-focused platforms have historically been harder to build large businesses around than modality-focused ones. As seen in the table above, the only truly stand out discovery platforms in history are the pioneer of rational drug design and of utilizing genetic evidence.
Both turned out to be the rare paradigm shift in the process and efficacy of drug development. Nowadays, developing drugs without any semblance of rational design feels prehistoric. Meanwhile, genetic evidence for drug targets has proven 2x the odds of clinical success with clear causal genes (Mendelian traits and GWAS associations linked to coding variants) being even higher!
To frame the rarity of truly paradigm-shifting discovery processes, consider that the number of novel targets approved by the FDA each year is usually between two and four.
The reason for this historically greater difficulty might lie in the difficulty, long duration, and capital intensity of getting a novel discovery process to a minimally disruptive product and product-disease fit. As such discovery platforms start off as point solutions, it takes heightened clarity to get to a disruptive process within a couple years.
Put more bluntly, it’s historically been easier to commercialize a novel modality capable of expanding the targetable space than to reinvent the scientific process itself.
Whereas, modality platform biotechs are selling end products not information to pharma. As Steven Holtzman puts it:
The output of Discovery Platform Companies is data/information/insights, NOT, as with the Modality Companies, New Chemical Entities and biologic therapeutics. The history of data in the biopharmaceutical industry is the history of its commoditization. Companies whose “life’s blood” is drugs/products have a vested interest in rendering data “pre-competitive” (or, at least doing so after they have had proprietary access for a time). They win based on their products; they don’t want to be held captive by the owner of the information. A more restrictive intellectual property (IP) environment: gone are the days when a transcriptional profile showing over-expression of a gene in a diseased tissue (or a genetic mutation in the diseased state) could get you an issued patent of the logical form, “a method of treating disease X comprising modulating target A by any means” (with dependent claims stating that the “means” could be an antibody, an antisense, an RNAi, a gene therapy, a small molecule, etc.). Moreover, the demands of the customer base became more expansive over time. Whereas, in the 1990s, most big pharma customers were willing to agree to terms that restricted their use of the data to the discovery and development of small molecule drugs (because that is all they did), in the present all pharmas/big biotechs would demand the right to exploit the data for all therapeutic modalities.
Because pharma is reluctant to pay more than minuscule amounts for pre-clinical information or change their workflow around novel technology, discovery-focused platforms tend to adopt a JV-like business strategy of orchestrating their own tech to help pharma hit difficult targets. It also means that discovery platforms tend to need to move faster up the value chain into an integrated internal pipeline than modality companies.
This industry dynamic of pharma paying pennies for pre-clinic discovery tools may change if and only if pharma starts to view novel approaches as existential threats. We’re currently seeing this play out with our portfolio company Wayve. Just within the last year, the legacy car OEMs have internalized that AVs are here and are existential to their very survival. Negotiations have shifted from contracts for tiny margins won via hand-to-hand combat over 2-year sales cycles to gargantuan deals over 3 month sales cycles.
We at Compound expect these problems will be increasingly alleviated under the theses that:
We are exhilarated by the dozens of emerging technologies to bend the probabilities of discovery and have been investing in many different versions.
As Holtzman framed Millennium’s pioneering of the discovery platform business model:
In reality, using large partnerships to create out-size value requires a keen sense of the external partnering environment, identifying the potential partner(s) which at that specific moment in its/their history has/have come to perceive a critical need for what you have, and crafting deals in which you, while you sell rights, retain the potential for value creation by, for example, retaining ownership of the knowledge. That, not a cookie-cutter model, is your legacy in this arena.
Broad vs Deep Discovery Platforms
Finally, not all insights platforms are the same: technologies with broad applicability to many different biological pathways and drug targets—like Millennium with genomics. Second, there are platforms with a deep disease-specific focus. One of the most successful examples is Agios, which primarily focuses on metabolic insights into cancer. The narrower the focus for an insights platform, the more its strategy starts to resemble a product-focused company. There are less “shots on goal,” so each one needs to count. As a result, they realistically need to land a sizable multi-target “foundational corporate partnership” earlier on—like Agios did with Celgene—and then push even harder to advance their own internal programs.
Building Platforms More Broadly
Over the last 10+ years, the dominant meta in biotech has swung violently and irrationally between being all-in on maximally general platforms and conservative single assets plays.
We at Compound push back on the recently popularized platform playbook of raising hundreds of millions, then taking 5+ years to build out the most generalized tech infrastructure possible, only after that start thinking about what target/disease to apply it to, and then pursue a pipeline of 10+ drug candidates under the flawed logic of maintaining maximal optionality (which just means that no candidate will receive adequate focus).
The reality is that no matter what, ultimately all platforms:


Moreover, nearly 50% of biotechs go bankrupt due to lack of continued funding vs just 33% for clinical failure. This further accentuates the misguided rationale for maximally general, long duration infra buildout without targeted end-points in mind.
We at Compound focus our bio investing efforts on how to build platforms thoughtfully. We firmly believe that platforms should be built in a step-wise, capital constrained / gated way with an intense focus on building the initial technology towards a specific target/disease uniquely unlocked by your technology.
Not coincidentally, the rough playbook above is how all the greatest biotech platforms in history were built.
Non-dilutive capital from early pharma partnerships funds further investments into the platform → making it more capable → making odds of drug development success tick up and/or making it useful to more researchers and targets → drawing more non-dilutive opportunities → meanwhile, partnering with pharma gives the team a first-hand look at drug development → strengthening the case for internal pipeline development, which is the way to capture the value you create.

Millennium got acquired for $9B off of only $8M in VC funding (despite building out custom wet lab automation equipment, assays, etc. which required $50M in annual CAPEX by its third year).
To execute this playbook, it’s essential to have a strong idea of which potential pharma partners have a strategic imperative to succeed in your focus areas, because those are the only entities with whom you’ll have the pricing power to earn a nice premium for all your efforts.

Lastly, our team at Compound gathered clinical, partnerships, and financial data on the 90+ most successful platform biotechs of all time to more precisely understand what these companies typically look like as they scale. Please treat the results as illustrative, not as fact.







Deal Structure Heuristics



"The human population, through explosive growth, has performed a comprehensive saturation mutagenesis experiment on itself. It is now the case that any single base substitution that is compatible with life is expected to be present somewhere among the nearly 8 billion living humans. Humanity has thus, in effect, done many of the natural experiments required to understand our own genotype-phenotype map; this leaves geneticists to catalog the outcomes of those experiments, and to leverage both observational and experimental approaches to understand the mechanisms by which variants alter biology."

https://leadershipandbiotechnology.blogspot.com/2018/08/early-stage-biotech-value-creation_15.html
https://www.bio.org/clinical-development-success-rates-and-contributing-factors-2011-2020
https://shelbyann.substack.com/p/a-playbook-for-human-evidence
https://www.mackenziemorehead.com/autonomous-science-part-i-everythings-an-api-away/
https://shelbyann.substack.com/p/commercializing-autonomous-science
https://www.mackenziemorehead.com/the-fickleness-of-scaling-laws/
Lessons learned from the fate of AstraZeneca's drug pipeline: a five-dimensional framework
https://rapport.racap.com/all-stories/semper-maior-2026-biotech-ma
https://www.science.org/doi/10.1126/science.abi8207
https://shelbyann.substack.com/p/predicting-protein-dynamics-moats
https://www.mackenziemorehead.com/untitled-2/
https://reconstrategy.com/2025/04/preclinical-licensing-deals-realized-value/
https://centuryofbio.com/p/on-biotech-platform-strategy
https://www.michaeldempsey.me/blog/2025/10/03/sequencing-vs-equal-odds-applied-research/
The Entrepreneur’s Guide to a Biotech Startup https://ott.emory.edu/_includes/documents/sections/startups/guide_to_biotech_startup.pdf
Company Histories
Physics / computation-first small-molecule design
Fragment-based lead discovery
DNA-encoded libraries / DNA-templated chemistry
Chemoproteomics & ligandability mapping
Phenotypic / cell-state modeling
Human genetics / in-human functional genomics–first
Antibody & binder discovery platforms
Natural-products, genome mining & metabolomics-first
Hotspot/allostery & dynamic pocket discovery
https://www.youtube.com/watch?v=IFbJIV8Sidw&ab_channel=NewLimit
https://centuryofbio.com/p/manifold
